Here, we utilize an electrochemical flow cell combined with an inductivelyĬoupled plasma mass spectrometer (ICP-MS) to enable the in situ study Photoelectrochemical activity of transparent semiconducting oxides. Photocorrosion can be significant and plays a central role during Is central to various areas, including design of catalysts and catalysisĬonditions, as well as passivation of metal surfaces. 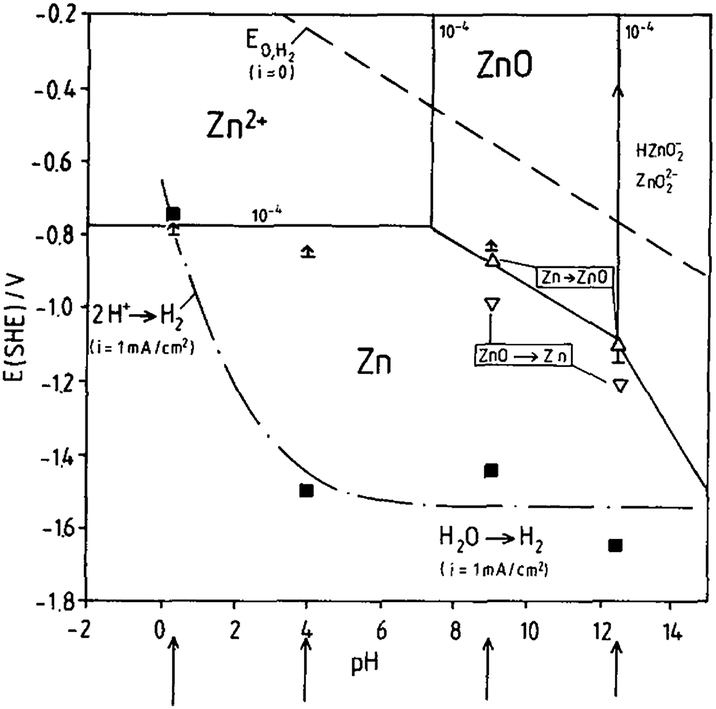
Our data demonstrates that surface design and fundamental understanding of surface chemistry provides an effective path to rendering electroactive surfaces stable under operating conditions.Īnd dissolution of transparent semiconducting oxides Whereas ZnO(101̅0) sustains higher dissolution rates, while it is inactive for water splitting. In particular, polar ZnO(0001) shows ideal stability at low potentials and under hydrogen evolution conditions. The observed dissolution behavior agrees well with the surface chemistry and stabilization mechanism of ZnO surfaces. A significant influence of the UV radiation and the pH of the electrolyte was observed. At acidic and alkaline pH, we characterized potential dependent dissolution rates into both the oxygen and the hydrogen evolving conditions. With this system we study the dissolution of zinc oxide single crystals with (0001) and (101̅0) orientations. Here, we utilize an electrochemical flow cell combined with an inductively coupled plasma mass spectrometer (ICP-MS) to enable the in situ study of the time-resolved release of zinc into solution under simultaneous radiation of UV-light. In particular, photocorrosion can be significant and plays a central role during photoelectrochemical activity of transparent semiconducting oxides. Degradation and dissolution of transparent semiconducting oxides is central to various areas, including design of catalysts and catalysis conditions, as well as passivation of metal surfaces. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
